波普医药正在热销:
别名:溴甲基环丙烷
英文名称:英文名称:(bromomethyl)cyclopropane
分子式:c4h7br
cas no.:7051-34-5
分子量:135
结构式: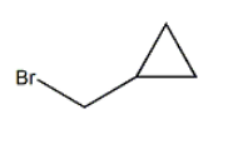
规格:97%
用途:基础原料、有机中间体、医药中间体
合成工艺如下:
bromine; triphenylphosphine; n-methylacetamide;
reactants are commercially availlable.
bayer aktiengesellschaft, us6008420, a1, (1999) example 1 (for comparison) 1250 ml of dimethylformamide were placed into the reaction vessel, and then 280.8 g of triphenylphosphine and then 70 g of hydroxymethylcyclopropane were added, the mixture was stirred for 30 minutes at room temperature under a nitrogen atmosphere, and the solution was sub sequently cooled to -10°c. then, 158.3 g of bromine (that is to say 2 mol percent more than theoret ically required) were metered in in the course of 4 hours. the reaction mixture was worked up by distillation. bromomethylcyclopropane was obtained in a yield of 77.5percent of theory. the purity of the product was over 97percent, the open-chain halogenoalkanes amounted to 0.6percent.
